Legionella remediation and Biofilm penetration
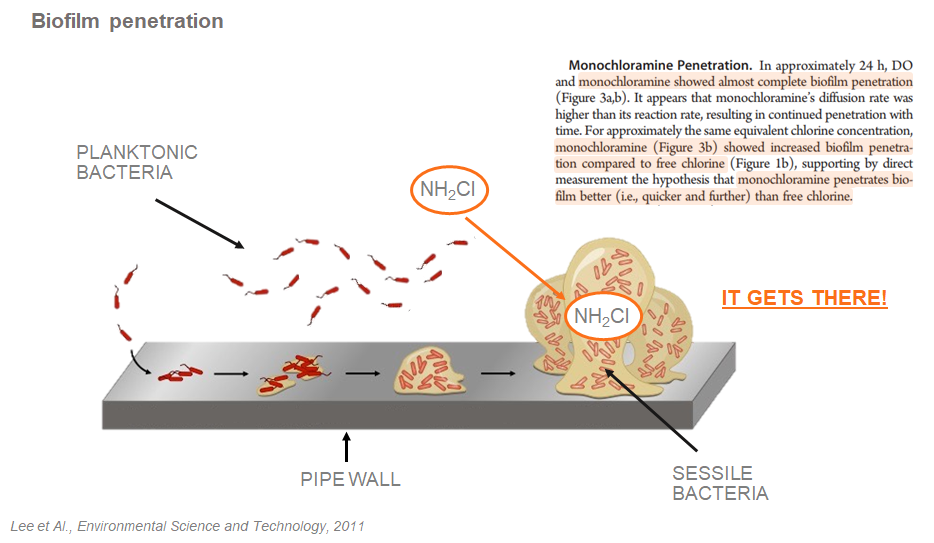
Thanks to its stability, monochloramine is the only disinfectant that can penetrate the biofilm.
What is Biofilm?
The biofilm is a slimy polymeric organic matrix that is attached to the internal pipe walls of a building water system. The biofilm is the place where bacteria, including waterborne pathogens, grow and start to colonize the plumbing system. The biofilm can be defined as a “city of microbes”.
There are two types of bacteria in a plumbing system: planktonic and sessile. The planktonic population includes all the bacteria that are not attached or inside the biofilm and that flow within the water stream. The sessile bacteria are the ones that are into the biofilm itself.
Water risk
Part of the biofilm and the sessile bacteria can detach from the pipe wall and by flowing within the water eventually colonize other parts of the building plumbing system, fixtures, faucets, and showers.
It is easy to understand that to achieve a complete disinfection an oxidizer needs to be able to penetrate the biofilm and get rid of both the planktonic and sessile bacteria.
Legionella remediation and Biofilm penetration: monochloramine does it better
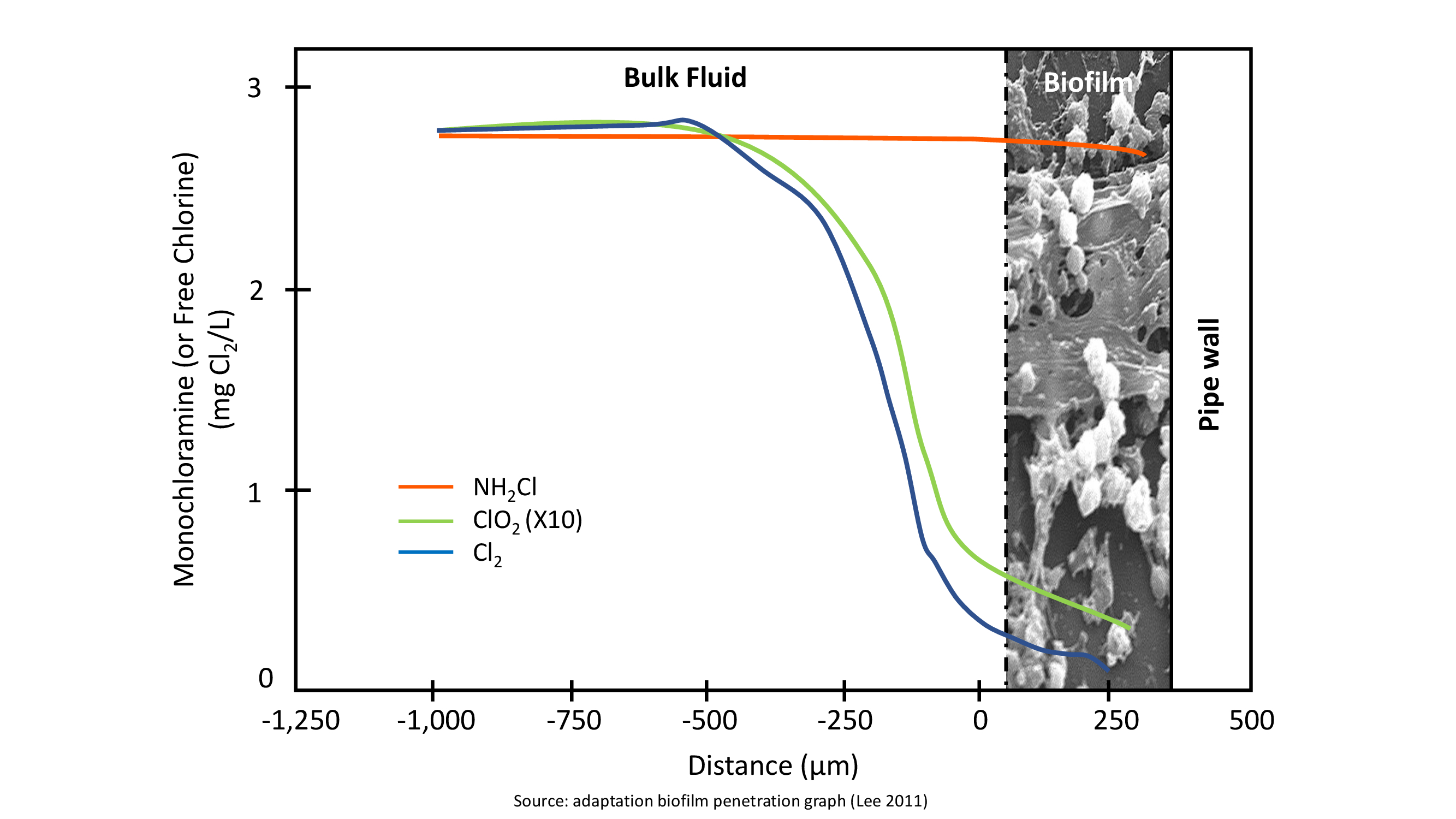
Because of their high reactivity traditional chlorine and chlorine dioxide rapidly react with the organic compounds of the biofilm with the result of having insufficient concentrations of biocide into the biofilm. In other words, chlorine and chlorine dioxide might be able to kill planktonic bacteria but they have no effect on the sessile population.
Being more stable, monochloramine is the only molecule that can penetrate the biofilm. SANIKILL monochloramine does not react with the organic compounds in the biofilm, and it is able to carry a consistent residual all the way inside and underneath the biofilm. This is the main reason why SANIKILL monochloramine is the best available supplemental disinfectant: because IT GETS THERE!